Constrictive Pericarditis or Restrictive Cardiomyopathy? The Echo Findings That Separate Them
One patient goes to surgery. The other does not.
Both can devastate cardiac function with a preserved ejection fraction and normal-looking walls.
One is surgically curable. The other is not.
Getting this wrong changes the outcome.
These two conditions sit at one of the hardest diagnostic crossroads in echocardiography. Here are the pivot points that separate them when everything else looks the same.
A few weeks ago we covered constrictive pericarditis step by step, and earlier this week we broke down restrictive cardiomyopathy in detail. If you haven’t read those yet, start there. The findings below will make a lot more sense with that foundation.
Why This Matters
The echo findings in CP and RCM overlap significantly. Both present with preserved ejection fraction, diastolic dysfunction, biatrial enlargement, and signs of right heart failure. But constrictive pericarditis is potentially curable with pericardiectomy, the surgical removal of the pericardium. Restrictive cardiomyopathy is not. In advanced cases, transplant is the only option. This distinction has to be made correctly.
The Pivot Points
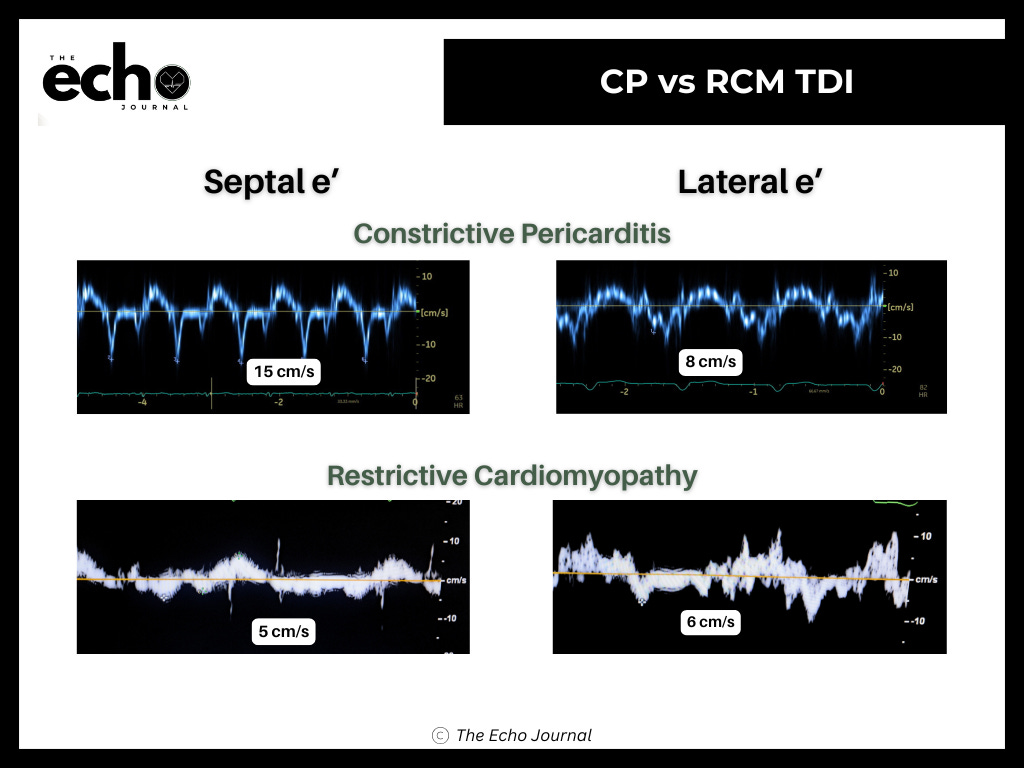
1. Tissue Doppler: Start Here
This is your most reliable separator.
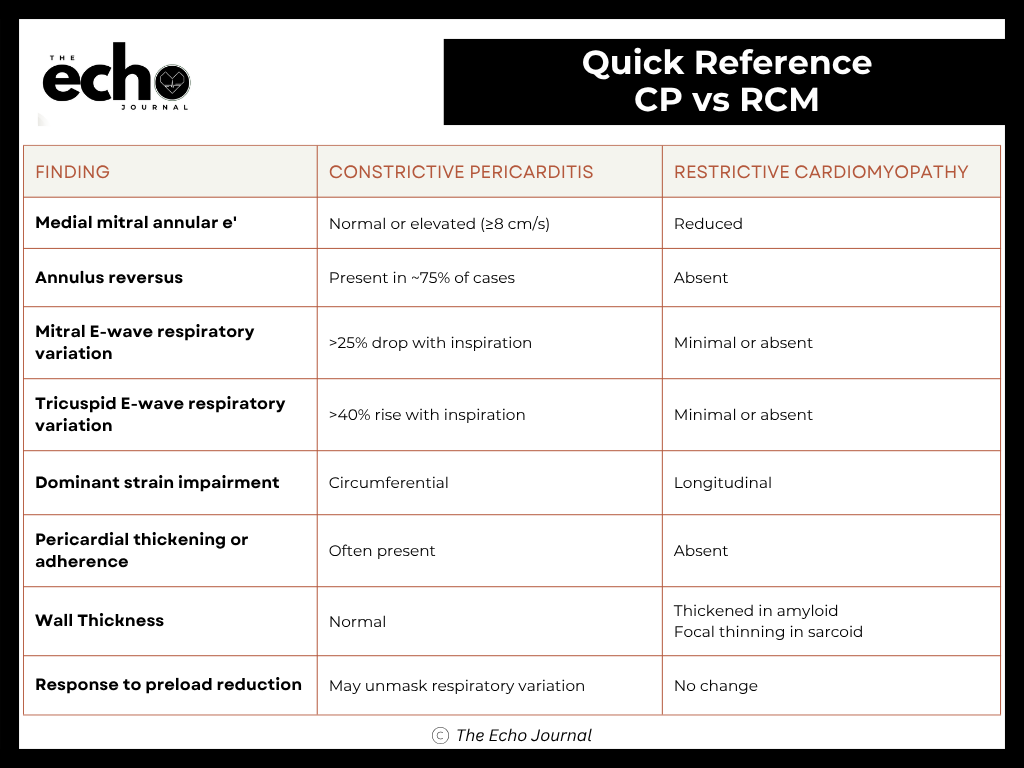
In RCM, the myocardium is stiff and longitudinal function suffers. The early diastolic mitral annular velocity (e’), a tissue Doppler measurement of annular motion, is reduced.
In CP, the muscle is often relatively preserved. The annulus hypermoves to compensate for constrained filling. Per ASE guidelines, a medial mitral annular e’ of 8 cm/s or greater in a heart failure patient should immediately raise suspicion for CP. This is called annulus paradoxus: a preserved or elevated e’ where you would expect it to be low.
The second clue is annulus reversus. Normally, lateral e’ exceeds medial e’. In CP, the fibrotic pericardium tethers the lateral wall and blunts its motion, so medial e’ equals or exceeds lateral e’. This reversal is present in up to 75% of surgically proven CP cases. It does not occur in RCM.
When medial e’ is higher than lateral e’, the pericardium is the most likely culprit. That pattern does not happen in myocardial disease.
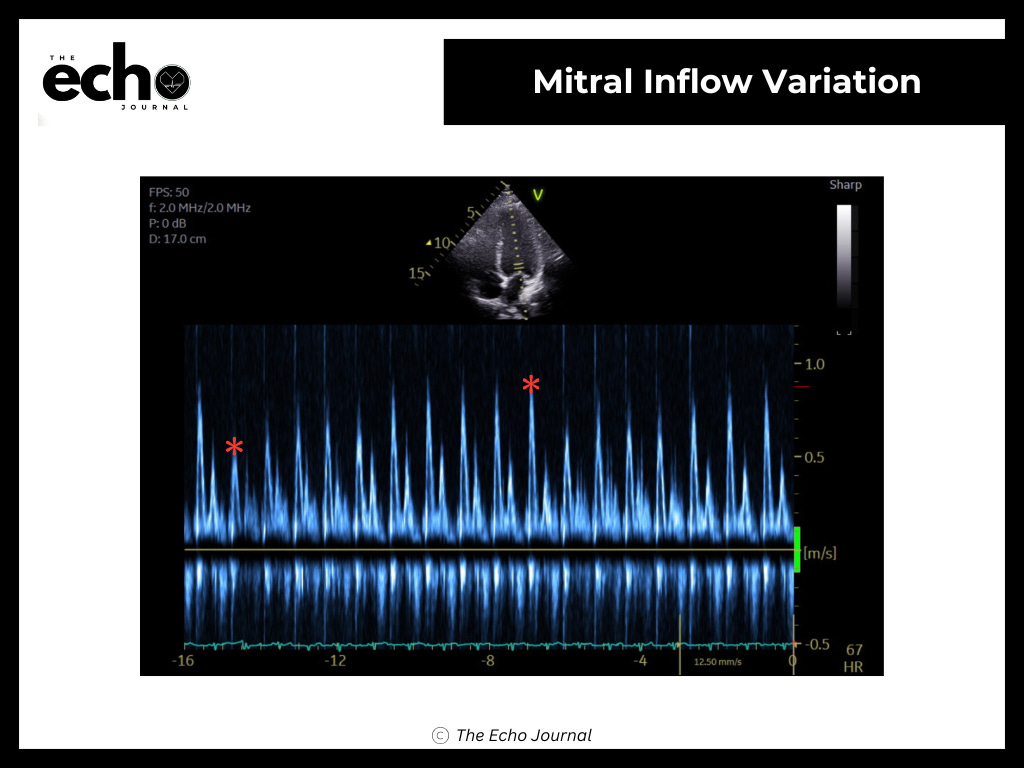
2. Respiratory Variation
In CP, the rigid pericardium creates exaggerated ventricular interdependence. When LV filling drops with inspiration, RV filling surges to compensate. The Doppler signature is specific: mitral E-wave velocity drops more than 25% with the first beat of inspiration, while tricuspid E-wave velocity increases more than 40%. That reciprocal seesaw is CP until proven otherwise.
In RCM, the pericardium is normal. Filling pressures are elevated equally across the respiratory cycle. Respiratory variation is absent or minimal.
One caveat: markedly elevated left atrial pressure can blunt this pattern in CP. If you suspect CP but are not seeing variation, reducing preload with a head-up tilt can unmask it. This has no effect in RCM.
3. Strain Pattern
GLS is reduced in both conditions, so the number alone does not help you. The pattern does. In RCM from infiltrative disease, particularly amyloidosis, longitudinal strain is significantly impaired, and you may see the characteristic apical sparing pattern. In CP, circumferential strain tends to be more affected than longitudinal strain. If the strain map looks like amyloid, RCM moves to the top of the list.
4. Pericardial Findings
If you can see pericardial abnormalities, they point toward CP. Look for a bright or thickened pericardium on 2D, loss of pericardial slide at the RV free wall, and a railroad track sign on M-mode. In RCM, the pericardium is normal.
A normal-appearing pericardium on echo does not rule out CP. CT and MRI are the gold standard for confirming pericardial thickness when the diagnosis is uncertain.
Constrictive Pericarditis vs Restrictive Cardiomyopathy
Key Takeaways
Tissue Doppler is your most reliable separator. A medial e’ of 8 cm/s or greater in a heart failure patient points toward CP.
Annulus reversus, where medial e’ equals or exceeds lateral e’, is present in up to 75% of surgically proven CP cases and does not occur in RCM.
Respiratory variation is the other key pivot. Mitral E-wave drop greater than 25% with inspiration and a reciprocal tricuspid rise greater than 40% support CP. Absent variation favors RCM.
Strain patterns differ: apical sparing and longitudinal impairment favor RCM, circumferential impairment favors CP.
Do not use E/e’ to estimate filling pressures in CP. Annulus paradoxus makes it unreliable.
References
Brandt, Roland R, and Jae K Oh. “Constrictive Pericarditis: Role of Echocardiography and Magnetic Resonance Imaging.” Escardio, 11 Nov. 2025, www.escardio.org/Journals/E-Journal-of-Cardiology-Practice/Volume-15/Constrictive-pericarditis-role-of-echocardiography-and-magnetic-resonance-imaging.
Garcia, Mario J. “Constrictive pericarditis versus restrictive cardiomyopathy?” Journal of the American College of Cardiology, vol. 67, no. 17, May 2016, pp. 2061–2076, https://doi.org/10.1016/j.jacc.2016.01.076.
Gerig, Don. “Constrictive Pericarditis vs Restrictive Cardiomyopathy - Echo Findings That Truly Differentiate.” Cardioserv, 6 Jan. 2026, www.cardioserv.net/constrictive-pericarditis-vs-restrictive-cardiomyopathy-echo-findings/.
Hatle, L K, et al. “Differentiation of constrictive pericarditis and restrictive cardiomyopathy by Doppler echocardiography.” Circulation, vol. 79, no. 2, Feb. 1989, pp. 357–370, https://doi.org/10.1161/01.cir.79.2.357.
Nagueh, Sherif F., Danita Y. Sanborn, et al. “Recommendations for the evaluation of left ventricular diastolic function by echocardiography and for heart failure with preserved ejection fraction diagnosis: An update from the American Society of Echocardiography.” Journal of the American Society of Echocardiography, vol. 38, no. 7, July 2025, pp. 537–569, https://doi.org/10.1016/j.echo.2025.03.011.
Nagueh, Sherif F., Otto A. Smiseth, et al. “Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American Society of Echocardiography and the European Association of cardiovascular imaging.” Journal of the American Society of Echocardiography, vol. 29, no. 4, Apr. 2016, pp. 277–314, https://doi.org/10.1016/j.echo.2016.01.011.
“Restrictive Cardiomyopathy.” Www.Heart.Org, 29 May 2024, www.heart.org/en/health-topics/cardiomyopathy/what-is-cardiomyopathy-in-adults/restrictive-cardiomyopathy.